A Summary of: Kava (Piper Methysticum) Extract for the Treatment of Nervous Anxiety, Tension and Restlessness.
Citation:
Kuchta, Kenny, Marie Hladikova, Michael Thomsen, Adolf Nahrstedt, and Mathias Schmidt. 2021. “Kava (Piper Methysticum) Extract for the Treatment of Nervous Anxiety, Tension and Restlessness.” Drug Research 71 (2): 83–93. https://doi.org/10.1055/a-1268-7135.
Quick Summary:
The study confirms the effective and safe short-term use of kava extract in the Commission E-defined indication of “nervous anxiety, tension and restlessness,” especially in other than chronic cases.
Full Summary:
The research paper entitled “Kava Extract for Situational Anxiety” serves as a groundbreaking investigation into the therapeutic efficacy and safety of Piper methysticum for treating symptoms commonly associated with nervous anxiety, tension, and restlessness. Conducted over a span of one year, from December 2000 to December 2001, the study was a multi-center endeavor involving 14 different research facilities and a total of 156 participants. The medication administered during the study was Kavasedon capsules, which contained a semi-liquid extract of the plant. Each capsule was standardized to contain 50 mg of kavalactones, the active compounds believed to be responsible for kava’s therapeutic effects.
The methodology employed in the study was rigorous and comprehensive. A five-item verbal rating scale was used to gauge the severity of 12 different symptoms commonly associated with anxiety disorders. These symptoms ranged from nervous tension and restlessness to more severe manifestations like panic attacks. The findings of the study were overwhelmingly positive, indicating not just statistically significant but also clinically meaningful improvements in all the symptoms that were assessed. The most notable improvements were observed in the symptoms of nervous tension and restlessness, which are often the most debilitating aspects of anxiety disorders.
The median duration for treatment was noted to be 28 days, which is a relatively short period for observing significant clinical improvements in symptoms of anxiety. This suggests that this extract could be an effective short-term treatment option for situational anxiety, which is often triggered by specific events or circumstances. The safety profile of kava was also robustly established through this study. Only one non-serious and reversible adverse event was reported, which was an increase in dreaming during the initial days of treatment. This is particularly noteworthy because one of the major concerns surrounding the use of kava has been its potential for causing liver toxicity. However, this study found no evidence to support such concerns, making it a significant contribution to the existing literature on it’s safety and efficacy.
One of the standout aspects of this study was its focus on the time required for the onset of kava’s therapeutic effects. According to the feedback provided by the study participants, the average time to experience the first noticeable effects of treatment was approximately 34.8 hours. This is relatively quick compared to other pharmacological treatments for anxiety, which often require weeks to show significant improvements. Interestingly, the study also found that patients with a shorter history of symptoms responded more favorably to kava treatment. This suggests that extracts might be particularly effective for treating acute episodes of anxiety, rather than chronic conditions.
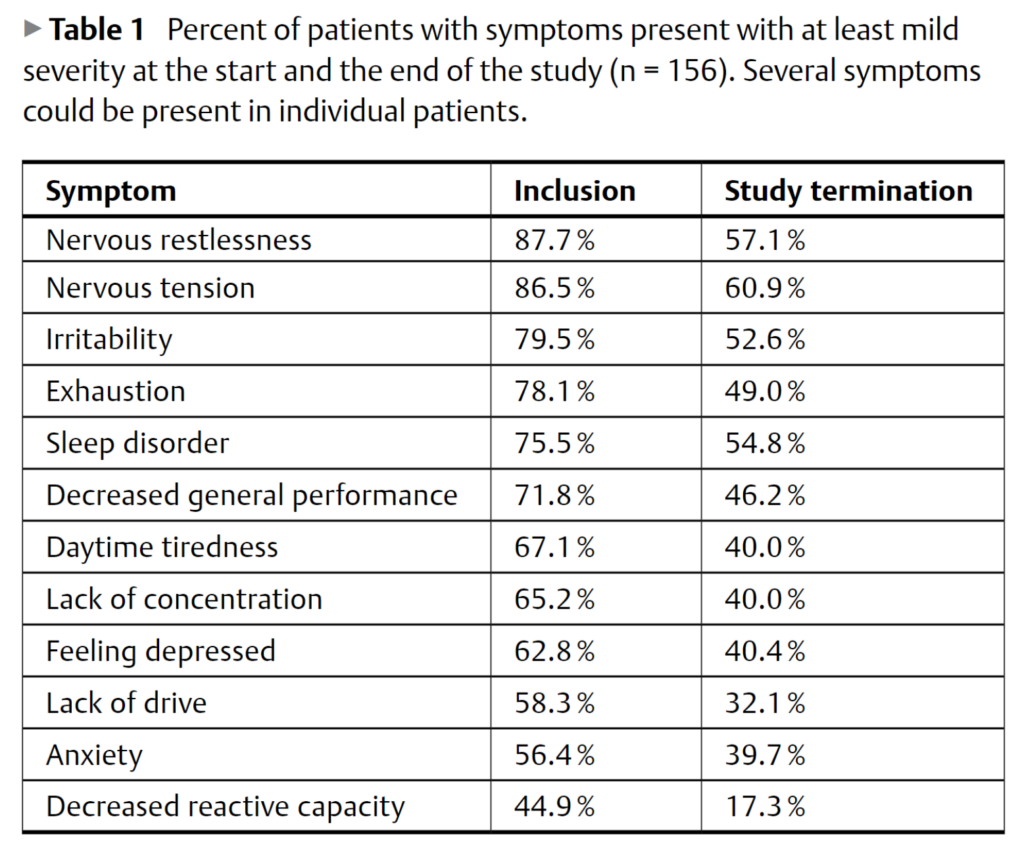
The timing of this study is also of critical importance. It was conducted during a period when kava was facing significant regulatory scrutiny due to concerns about its safety, particularly its potential for causing liver toxicity. Despite these challenges, the study did not find any substantial evidence to suggest that its use in treatment could lead to significant adverse effects. This makes the study an invaluable addition to the growing body of research that supports the therapeutic benefits of kava for treating anxiety-related conditions.
In conclusion, this comprehensive study not only substantiates the effectiveness of kava in alleviating symptoms of nervous anxiety, tension, and restlessness but also strongly endorses its safety. The study serves as a compelling piece of evidence for healthcare providers and patients alike, who are exploring alternative treatment options for anxiety-related conditions. It fills a significant gap in the existing literature by providing robust clinical evidence to support the use of kava as a safe and effective treatment for situational anxiety. Given the increasing prevalence of anxiety disorders worldwide, the findings of this study could have far-reaching implications for the future of mental health treatment.
The study, therefore, serves as a cornerstone in the scientific understanding of kava’s role in mental health, particularly in the treatment of anxiety disorders. It opens up new avenues for future research, including long-term studies to assess the safety and efficacy of kava for chronic conditions. It also sets the stage for more in-depth investigations into the mechanisms of action of kava’s active compounds, which could lead to the development of more targeted and effective treatments for a range of anxiety-related conditions.
Notes:
- The study spanned from December 2000 to December 2001.
- Kavasedon capsules were the medication used in the study.
- The median duration for kava treatment was 28 days.
- Only one non-serious, reversible adverse event was reported: increased dreaming during the initial days of treatment.

